Our portfolio company Sleepiz won a 50’000 CHF prize (including alumni contribution) at this year’s 30th anniversary edition of the W. A. De Vigier Foundation startup competition. Congratulations to Soumya and and the entire Sleepiz team! About Sleepiz Untreated sleep apnea patients suffer from a multitude of diurnal and nocturnal symptoms, such as loud snoring, […]
At the Swiss Diagnostics Startup Day in Olten, our portfolio company BioReperia won the 1st prize of the jury and the 2nd prize awarded by an audience of industry professionals. Congratulations to the winner of the Swissbiolabs Challenge 2019, Dr. Anna Fahlgren, runner-ups Marc Moghbel and Karim Brandt and 3rd place Dr. Christian Vogler for […]
Danuta Cichocka of HEMEX’s portfolio company Resistell AG had 90 seconds to explain what she does and how much money she needs to take her startup to the next level. She was one of four up-and-coming entrepreneurs who made the best pitch at the annual Startup Champions Seed Night. To the video
At the Start Summit held last week in St. Gallen, Sleepiz was crowned the Start Summiteer 2019. The start-up develops a solution helping more than 1 billion people with sleep disorders. Last week the start summit in St. Gallen brought together 3000 players in the startup ecosystem – from students to startups, investors, corporates and […]
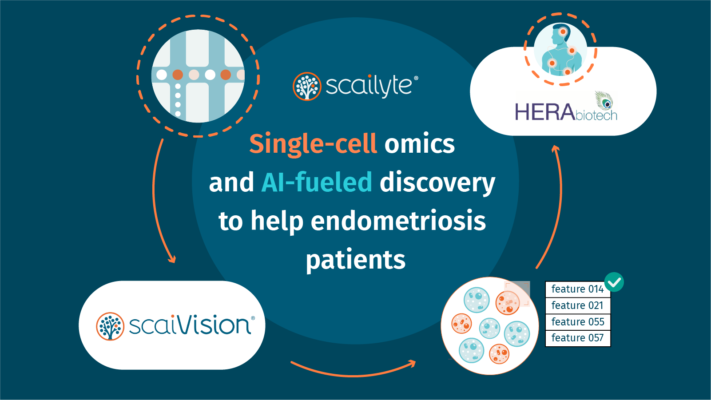
Scailyte AG, a Swiss startup in the emerging and rapidly growing field of single-cell technologies, raised CHF 2.75M in seed funding and gained Swisscom Ventures as a strategic investor. Zürcher Kantonalbank also joined the financing round, alongside the existing shareholders, thus reaffirming their trust in the competence and vision of Scailyte’s team. Dominique Megret, Head […]
Marija Plodinec, CEO of Artidis (Img: Technologiepark Basel) “Artidis is based on the oldest diagnostic test on earth: palpation,” says Marija Plodinec, CEO and co-founder of Artidis AG. “In a nutshell, we palpate tissue on a molecular level,“ she adds with a twinkle. That’s because instead of a physician’s hand, Artidis runs a fine, high-precision […]
Press release: Resistell AG closes seed round Resistell AG has today closed an oversubscribed seed financing round. Lead Investor Occident Group AG, Hemex AG and six other private investors invested in the EPFL spin-off developing the world’s fastest phenotypic antibiotic susceptibility test. 700,000 people die each year due to drug resistant infections. Unless we can […]
Scailyte develops artificial intelligence technology to enable precision medicine and early detection of complex diseases. The company has won the Start JLM competition recently and will explore and experience Jerusalem’s vibrant startup ecosystem in November. Start JLM gives entrepreneurs the opportunity to connect and benefit from interaction with prominent local tech leaders in Jerusalem in a 7-day immersive […]